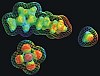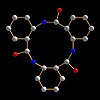
The highly unique crystal structure of nanotubes constructed from cyclic peptides is revealed this month by Japanese researchers in the journal Organic Biomolecular Chemistry. The descendents of these novel nanotubes could find a role in future molecular electronic devices, according to the team, who allude to the high macrodipole moment of their materials.
Shunsaku Kimura and colleagues at Kyoto University, have built on the work of ETH’s Dieter Seebach and Wisconsin’s Sam Gellman to use supramolecular chemistry to construct through self-assembly a stacked column of cyclic peptides, themselves made from three ACHC amino acids linked in a ring. ACHC is the trans-2-aminocyclohexylcarboxylic acid. The team used Fourier transform infra-red and nuclear magnetic resonance spectroscopy measurements and computational calculations to demonstrate that this cyclic tri-beta-peptide has C3-symmetry with the amide groups in the trans positions.
To read my full article visit the spectroscopyNOW.com crystallography channel.