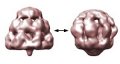
 A new study reveals that the static snapshots recorded in protein crystallography may be missing the bigger picture. Investigations of a bacterial protein using cryomicroscopy shows the protein in a balloon-like mode previously hidden from sold state studies. The discovery suggests that techniques complementary to X-ray crystallography are essential if molecular biology is to gain a complete understanding of protein structure.
A new study reveals that the static snapshots recorded in protein crystallography may be missing the bigger picture. Investigations of a bacterial protein using cryomicroscopy shows the protein in a balloon-like mode previously hidden from sold state studies. The discovery suggests that techniques complementary to X-ray crystallography are essential if molecular biology is to gain a complete understanding of protein structure.
Steven Ludtke, assistant professor of biochemistry and molecular biology and co-director of the National Center for Macromolecular Imaging at Baylor College of Medicine and colleagues Dong-Hua Chen and Wah Chiu there and Jiu-Li Song and David Chuang at The University of Texas Southwestern Medical Center in Dallas, studied a mutant protein and came to this perhaps not so startling conclusion. The protein GroEL chaperones misfolded proteins and nudges them into their active folded state in the cell. Protein misfolding is implicated in a number of neurodegenerative diseases, such as Alzheimer’s disease and the prion diseases including Creutzfeldt-Jakob disease.
The full story is now available at SpectroscopyNOW